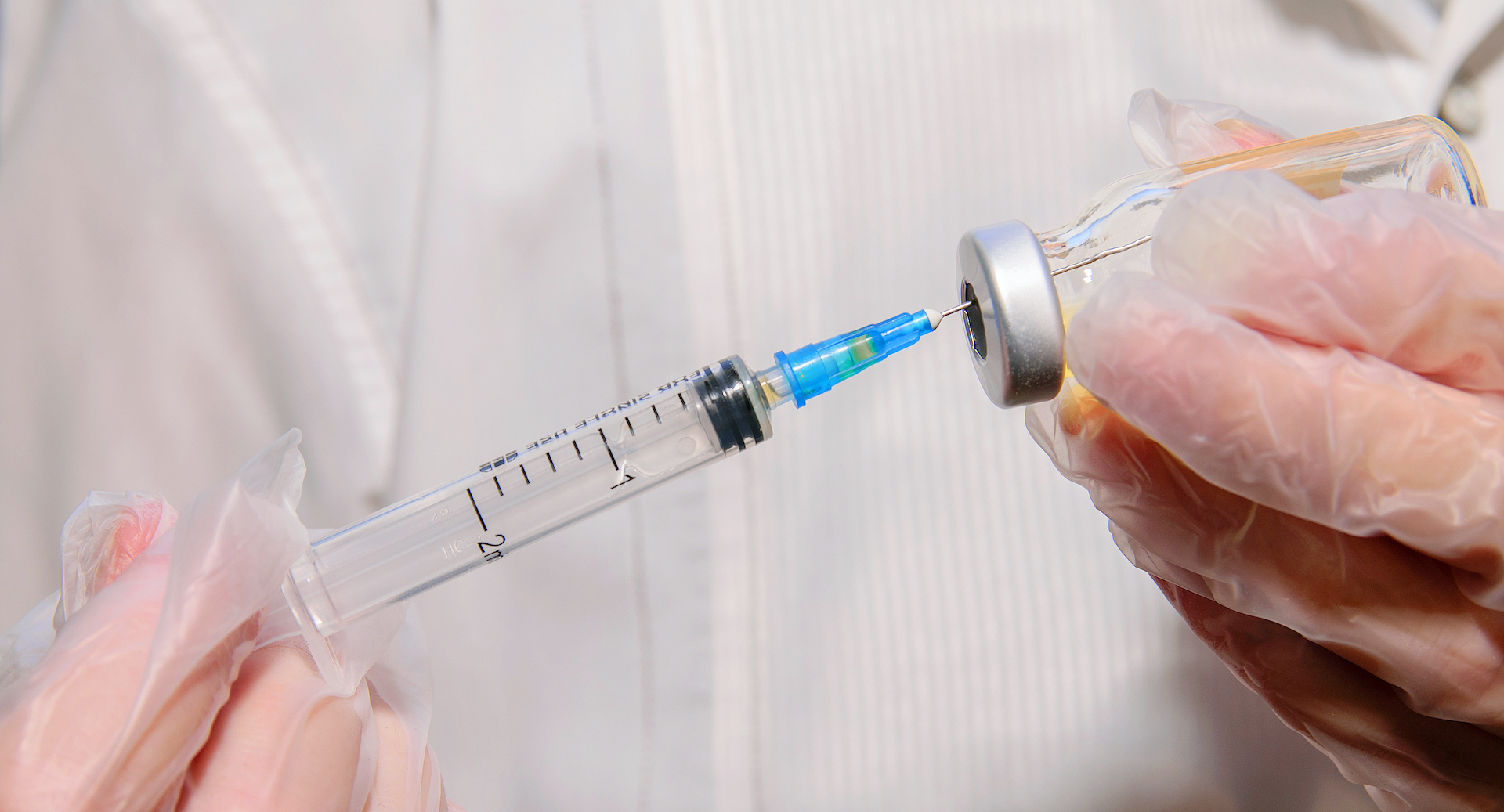
Hence then, the article about fda to authorize 3rd covid 19 vaccine dose for immunocompromised people was published today ( ) and is available on Live Science ( Middle East ) The editorial team at PressBee has edited and verified it, and it may have been modified, fully republished, or quoted. You can read and follow the updates of this news or article from its original source.
Read More Details
Finally We wish PressBee provided you with enough information of ( FDA to authorize 3rd COVID-19 vaccine dose for immunocompromised people )
Last updated :
Also on site :
- Today's NYT Mini Crossword Answers for April 29 – CNET
- Amazon’s $35 Seersucker Comforter Set Has a 'Cloud-Like Feel' That 'Makes You Want to Stay in Bed All Day'
- MI vs SRH Dream11 Prediction Today Match, Dream11 Team Today, Fantasy Cricket Tips, Playing XI, Pitch Report, Injury Update- IPL 2026, Match 41