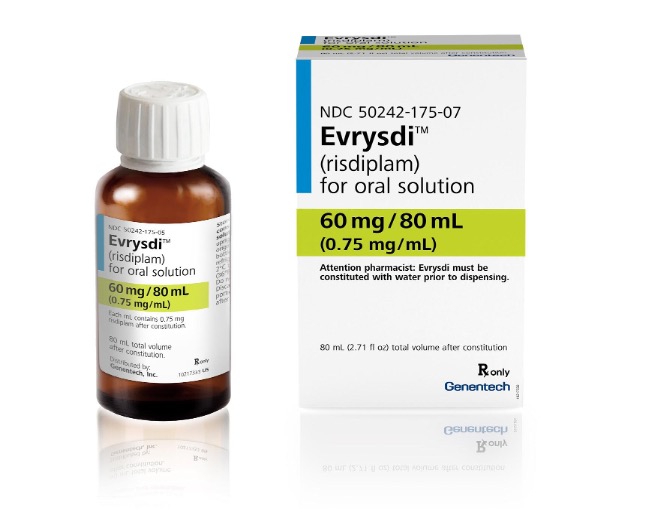
Hence then, the article about genentech recalls prescription drug evrysdi due to failure to meet child resistant packaging requirements risk of drug exposure through eye or skin absorption recall alert was published today ( ) and is available on CPSC ( Middle East ) The editorial team at PressBee has edited and verified it, and it may have been modified, fully republished, or quoted. You can read and follow the updates of this news or article from its original source.
Read More Details
Finally We wish PressBee provided you with enough information of ( Genentech Recalls Prescription Drug Evrysdi Due to Failure to Meet Child Resistant Packaging Requirements; Risk of Drug Exposure through Eye or Skin Absorption (Recall Alert) )
Also on site :
- PIN Stockholder Alert: Shareholder Rights Law Firm Robbins LLP Reminds Investors of the Class Action Lawsuit Against Pinterest, Inc.
- ‘Murphy Brown’ Star Jessie Jones Dead at 75
- 2026 APRA Music Awards Nominations: Full List