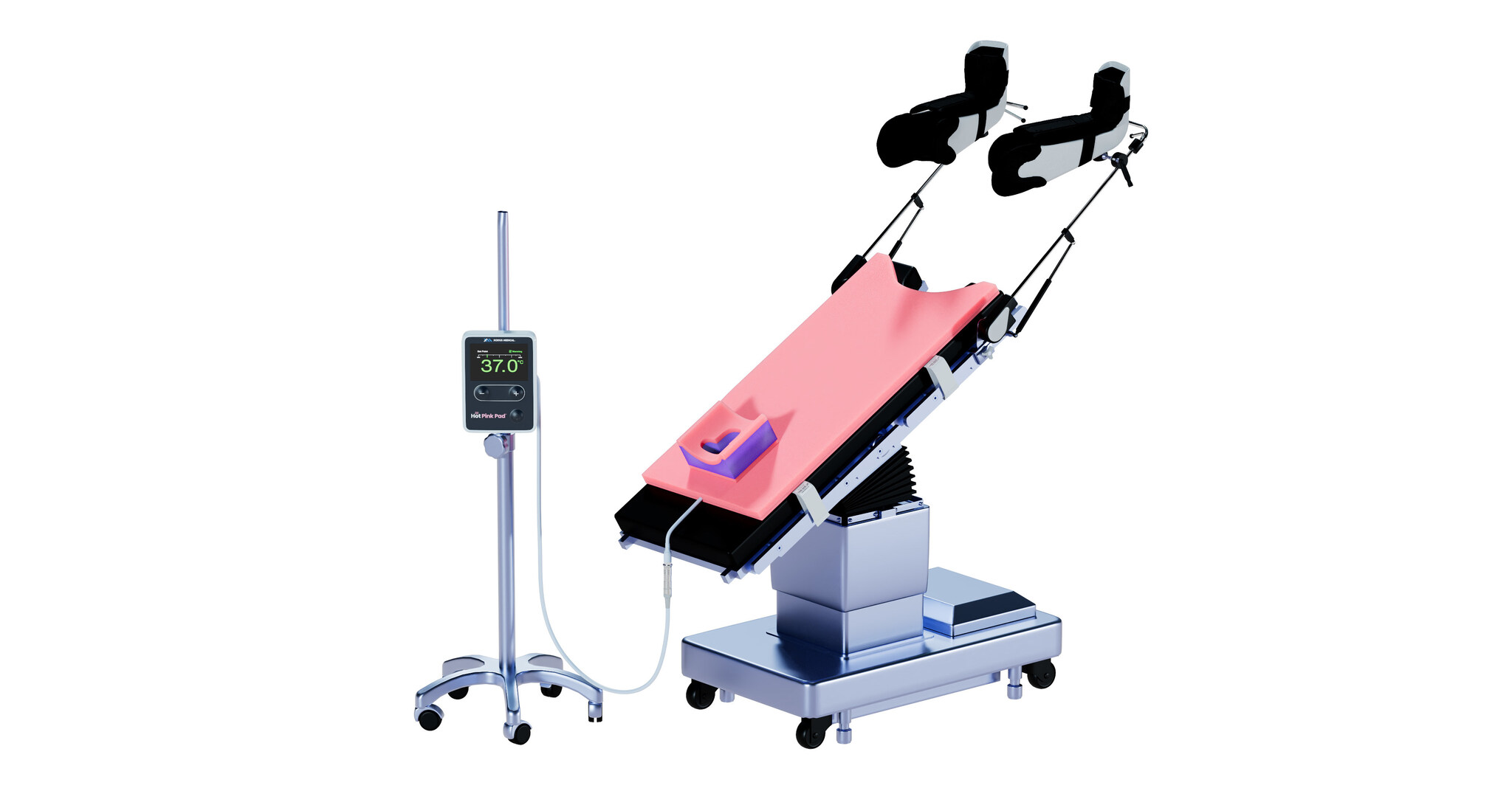
Integrated solution combines active warming and secure positioning to improve surgical outcomes and OR efficiency PITTSBURGH, April 17, 2026 /PRNewswire/ -- Xodus Medical today announced the launch of the Hot Pink Pad®, a next-generation patient temperature management and positioning...
Hence then, the article about xodus medical receives fda 510 k clearance launches the hot pink pad a breakthrough in surgical patient temperature management and positioning was published today ( ) and is available on PR Newswire ( Middle East ) The editorial team at PressBee has edited and verified it, and it may have been modified, fully republished, or quoted. You can read and follow the updates of this news or article from its original source.
Read More Details
Finally We wish PressBee provided you with enough information of ( Xodus Medical Receives FDA 510(K) Clearance, Launches the Hot Pink Pad®, a Breakthrough in Surgical Patient Temperature Management and Positioning )
Also on site :
- 57-year-old woman killed in Lake Geneva boat accident on as busy boating season begins
- Wind Advisory issued April 17 at 2:56AM PDT until April 17 at 8:00AM PDT by NWS Los Angeles/Oxnard CA
- Inside The ‘Landmark’ Exhibition Showcasing The Story of Black British Music at London’s Brand-New V&A