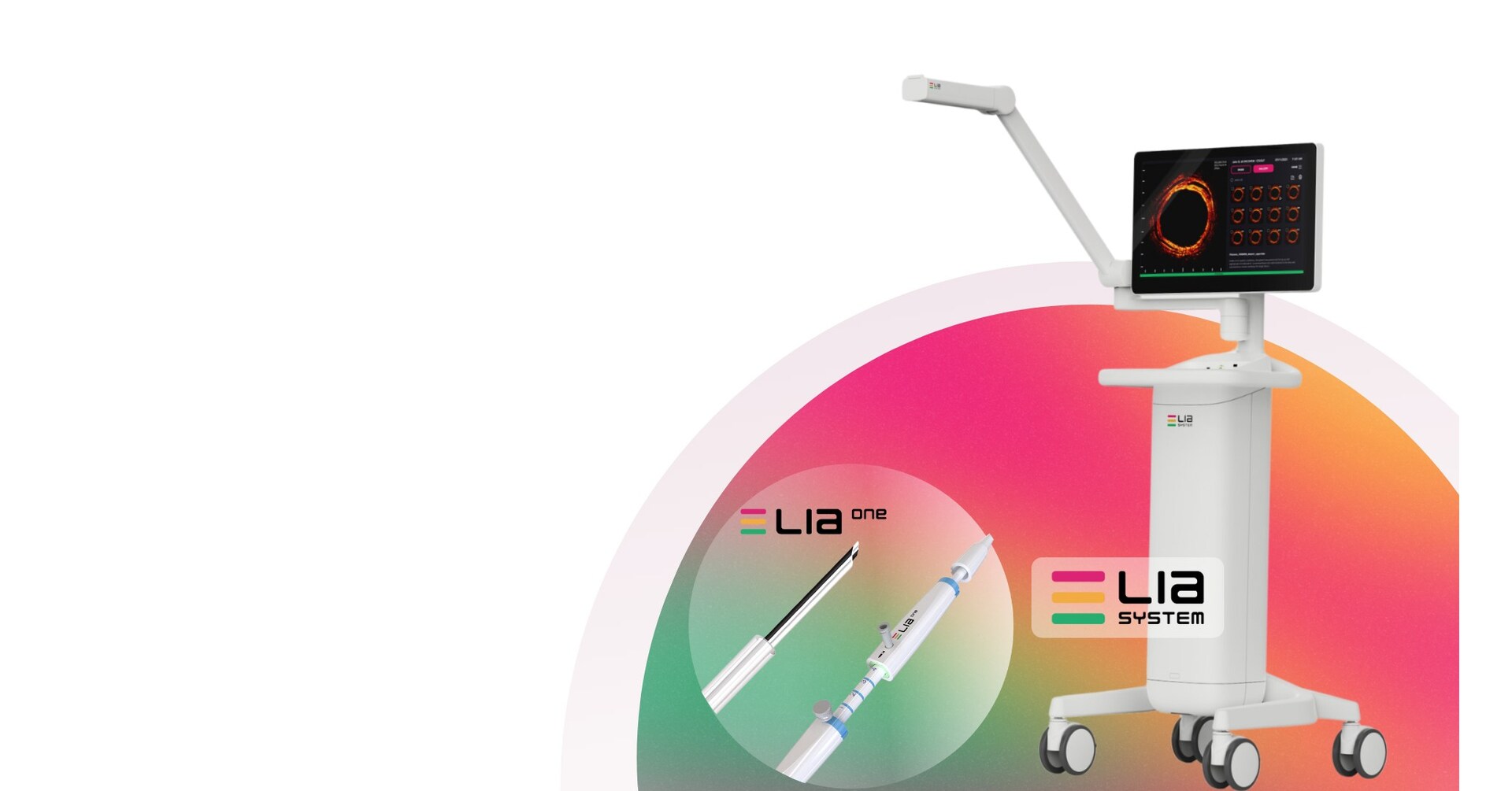
SAN JOSE, Calif., Feb. 5, 2026 /PRNewswire/ -- LEADOPTIK, Inc., a medical technology company focused on advancing precision lung cancer biopsy, today announced the successful first-in-human clinical use of its FDA-cleared Last Inch Assessment™ (LIA) System at University of Chicago Medical...
Hence then, the article about leadoptik announces first in human use of fda cleared lia system was published today ( ) and is available on PR Newswire ( Middle East ) The editorial team at PressBee has edited and verified it, and it may have been modified, fully republished, or quoted. You can read and follow the updates of this news or article from its original source.
Read More Details
Finally We wish PressBee provided you with enough information of ( LEADOPTIK Announces First-in-Human Use of FDA-Cleared LIA™ System )
Also on site :
- Fintech Media Advertising wird offizieller Sponsor des Honda LCR-Teams und festigt damit seine Position als globale Fintech-Infrastrukturplattform
- EduBlock Pro Wins STPI Award at TiECON Chandigarh 2026, Cementing Its Position as India's Most Trusted Blockchain-Powered Exam Integrity Platform
- How Iran, Suffering Under Sanctions, Diversified Its Economy