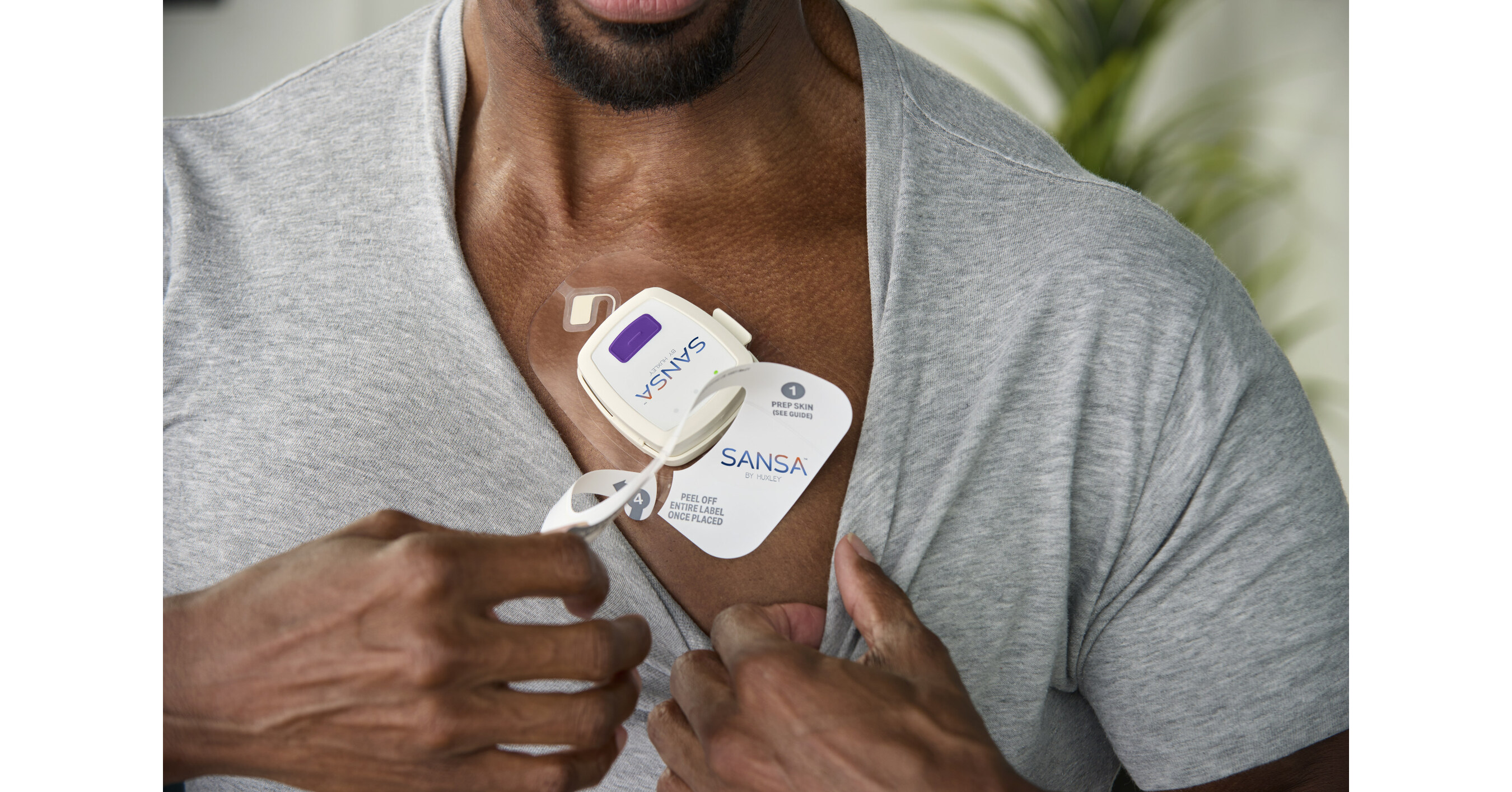
Hence then, the article about u s food and drug administration gives huxley medical 510 k clearance for sansa home sleep apnea test was published today ( ) and is available on PR Newswire ( Middle East ) The editorial team at PressBee has edited and verified it, and it may have been modified, fully republished, or quoted. You can read and follow the updates of this news or article from its original source.
Read More Details
Finally We wish PressBee provided you with enough information of ( U.S. Food and Drug Administration Gives Huxley Medical 510(k) Clearance for SANSA Home Sleep Apnea Test )
Last updated :
Also on site :
- Go Inside BTS ‘Tonight Show’ Taping, From Fan Jump-Scares to Jimmy Fallon Angling to Be ‘The 8th Member’
- Lord of the Rings Trilogy Has Many Elements That Are Difficult to Watch in the Modern Day
- Trump& 039;s Disapproval Rating Hits All-Time High, Fox News Poll Shows