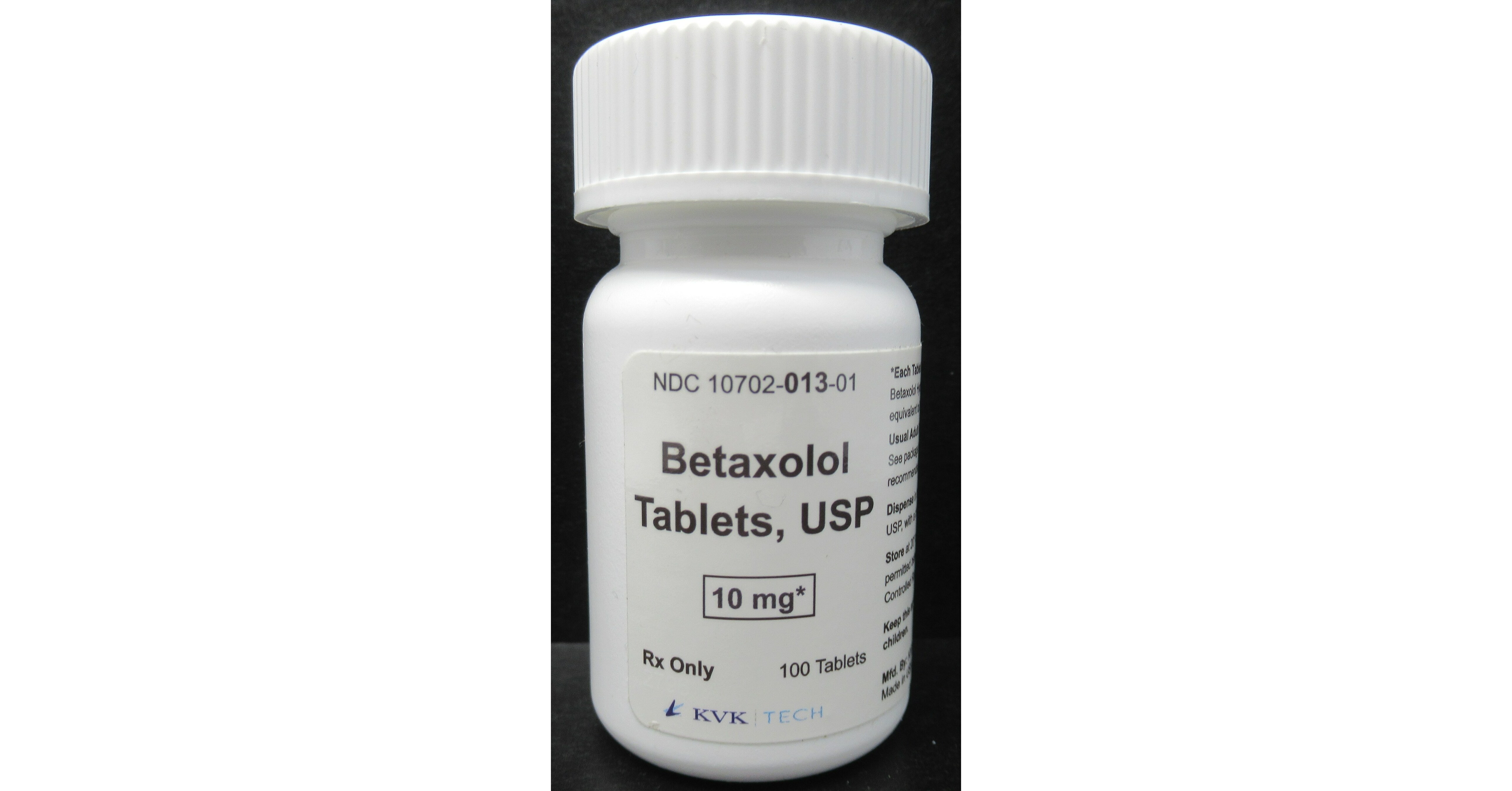
Hence then, the article about kvk tech inc issues voluntary nationwide recall of one lot of betaxolol tablets usp 10 mg batch number 17853a as a precautionary measure due to a single foreign tablet found during the line clearance after the batch was packaged was published today ( ) and is available on PR Newswire ( Middle East ) The editorial team at PressBee has edited and verified it, and it may have been modified, fully republished, or quoted. You can read and follow the updates of this news or article from its original source.
Read More Details
Finally We wish PressBee provided you with enough information of ( KVK-Tech, Inc. Issues Voluntary Nationwide Recall of one lot of Betaxolol Tablets, USP 10 mg (Batch Number: 17853A) as a precautionary measure due to a single foreign tablet found during the line clearance after the batch was packaged )
Also on site :
- Homemade dumplings are a Lunar New Year staple. Last week, a cafe invited those who’d never made them to give it a try.
- Yankees& 039; Giancarlo Stanton Reveals How Injury Impacts Everyday Life
- Vicky Oganyan coaching Burroughs, LA Mission College basketball to historic seasons